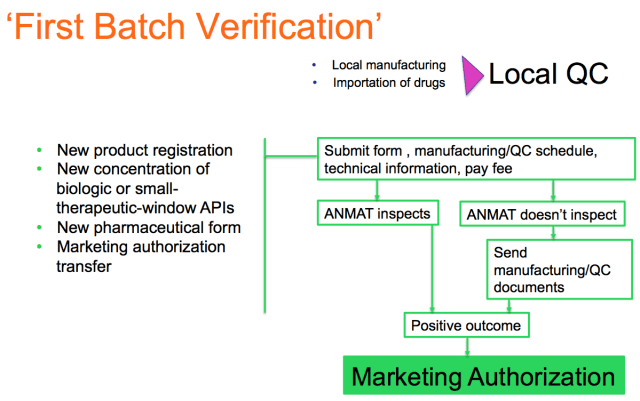
For the following cases:
- new product registration
- new concentration of biologic or small-therapeutic-window APIs
- new pharmaceutical form
- marketing authorization transfer
a procedure called first batch verification will take place.
Before the release of the first batch for commercial purposes, the manufacturing or importing company has to request the first batch verification, by submitting the corresponding form, the manufacturing and/or quality control schedule, the relevant technical information and by paying a fee.
ANMAT will then either physically inspect the manufacturing and/or control processes in the dates provided in the schedule, OR review the manufacturing and/or control records afterwards as a ‘documental verification’.
Upon a positive outcome, a marketing authorization will be granted.
Last update: October 2013




